Thanks to a boost in popularity over recent years, many of us will be aware of some the benefits of a plant-based or vegan diet, with more and more people becoming interested in, or taking up, this kind of lifestyle. The question is, how do these diets work in the world of sport and what affect does it have on athletes?
In our latest Nutrition X-Change, Dr. Nanci Guest - whose current post-doctoral work at the University of Toronto includes continued research in nutritional genomics and plant-based eating, looks into the influence of a vegan/plant-based diet on nutrition and athletic performance. Dr. Guest delves deep into the motivations behind following this kind of diet, considerations to bear in mind and the ergogenic aids that may be needed for an athlete to perform at their highest level.
Read the paper in full below or click the button to download as a PDF.
PLANT-BASED AND VEGAN DIETS IN EXERCISE AND SPORT
Practical Implications
- Whole foods plant-based diets are increasing in popularity as mounting evidence suggests that they decrease the risk of cardiovascular diseases, diabetes, cancer and all-cause mortality and also promote a healthier environment and help mitigate global warming.
- Motivations for following a plant-based diet may include improving physical health, reducing the environmental impact of food choices, animal rights/ethics, and in the case of athletes optimizing body composition and exercise and sport performance.
- As with omnivorous diets, there are certain nutrients for which athletes following a plant-based diet should be mindful of consuming in appropriate quantities.
- Although vitamin B-12 is the only nutrient that cannot be sourced exclusively from plants, nutrients to be mindful of on a plant-based diet include protein, omega-3 fats, iron, zinc, calcium, and vitamin D.
- Several studies show that plant-based eating patterns neither improve nor hinder physical performance or resistance-training goals, which may bring comfort to those committed to a vegan lifestyle for ethical reasons.
- Animal protein is not required to build and repair muscle since well-balanced plant-protein meals can provide an excess of all the essential amino acids with sufficient bioavailability.
- Select ergogenic aids (creatine, carnosine [via beta-alanine]) are required in supplement form to enhance performance in both omnivorous and plant-based athletes.
- Athletes who decide to transition to a diet that is mostly or exclusively plant-based can feel confident that they will typically see an improvement in their overall nutrient intake due to increased intakes of fruits, vegetables, beans, nuts and seeds, and whole grains that provide high levels of vitamins, minerals, antioxidants and fiber, while also being very low in saturated fat.
Introduction
Interest in the benefits of a plant-based (PB) diet has risen sharply in recent years. Accordingly, it is important to examine the impact of PB diets not only on physical health, but also on exercise capacity and performance in athletes and active individuals. Athletes’ dietary patterns range substantially in the degree of inclusion of animal-sourced foods (ASF), from high intakes to the complete avoidance of foods or food ingredients of animal origin. With many athletes making the “switch” to a PB diet, it is relevant to consider whether the replacement of ASF with plant proteins and plant-focused eating in general may impact exercise and sport performance.
Under the umbrella of “plant-based”, vegans and vegetarians share a disavowal of meat or animal flesh consumption but differ regarding the use of ASF (e.g., eggs) and animal by-products (e.g., casein). The term “plant-based” is sometimes used interchangeably with “vegan” to describe a dietary pattern that is 100% plant-based or exclusively derived from plants [1], but most individuals seem to understand “plant based” as being more like vegetarian, i.e., “mostly” plant-based. This review will use the term “plant based” to indicate that the diet is based on “all” or “almost all” plants. This implies that a serving of milk, cheese, eggs, fish or foods containing animal derived ingredients are consumed never or rarely, i.e., no more than 1-2 times per week. In comparison, the term “vegan” or “veganism” is characterized as a justice movement and lifestyle that not only includes a strict PB diet, but also condemns the exploitation and use of animals in any form (e.g., for use as food, clothing and or entertainment). Accordingly, all vegans consume a PB diet but not all PB eaters are vegan.
Factors contributing to the rise in popularity of PB diets include: (1) health benefits associated with increased intakes of plant-sourced proteins; (2) concerns regarding adverse health effects of consuming diets high in animal protein (e.g., increased saturated fat); 3) the impact of meat on climate change and the environment; and 4) ethical issues regarding the treatment of animals, including concerns for the increased risk of future pandemics through high-density livestock operations [2, 3]. For many athletes, however, the question is at what degree of restriction of animal foods will I see a performance benefit? But perhaps a more appropriate question to ask is “Can a plant-centered diet adequately serve my health, body composition and performance goals”? That is, can an athlete adopt a strictly PB diet and still expect to perform optimally? Beliefs and attitudes in the sporting community surrounding the need for animal-sourced proteins must embrace a conceptual paradigm shift if we are strictly basing nutritional strategies on how it relates to performance outcomes. In essence, a PB diet need not be superior to one including ASF, but it must be at least equivalent, which then makes it a viable and acceptable option for athletes.
This review will outline which nutrients may need more consideration when adopting a PB dietary pattern, how this may impact exercise performance and recovery, and address concerns framing the appropriacy of plant proteins for hypertrophy and strength. Concern for adequacy and bioavailability for plant-sourced micronutrients and the need for nutritional supplements for general health or ergogenic performance enhancement will also be discussed.
Plant-Based Diets, Health and Performance
The accumulating evidence on the health benefits of PB diets have made them of interest not only to consumers, but also to healthcare professionals and researchers. Among Western populations PB diets are being adopted for their role in optimizing health and the ability to lower the risk of many chronic diseases [4]. Although there is significant heterogeneity in the types and specific definitions of PB diets, the complete exclusion of animal products has been found to be significantly associated with beneficial changes to health. These beneficial changes mainly impact cardiometabolic CVD risk factors, through lower BMIs, improved blood glucose and lipid profiles, lowered inflammation, and blood pressure [1, 2, 5-9] , while also showing a reduced risk of several cancers [10-12] compared to most animal-heavy omnivorous diets, which are typically lower in whole grains, vegetables, fruits, nuts and seeds, and legumes [13].
Habitual dietary intake also has a profound effect on shaping the human microbiome which is not a fixed trait but instead responds to environmental stimuli such as nutrition, and is a malleable part of the human health. The benefits of plant-derived nutrition on the composition and diversity of the human gut microbiota have been noted after the adoption of a PB diet [14, 15]. For example, prebiotics are non-digestible dietary plant fibers that stimulate growth of beneficial gut bacteria and confer a health benefit upon the host [16]. Common food sources of prebiotics are pulses, artichokes, whole grains, bananas, peas, beans, asparagus, and garlic, leeks and onions [16]. Of note, high protein intakes (typical in athletes) without plant derived dietary fiber may offset the positive effect of training and exercise on gut microbiota but may also significantly lower the relative abundance and diversity of beneficial bacteria [17, 18]. Health promoting effects of the gut microbiota are primarily derived through the fermentation of dietary fiber to produce short chain fatty acids (SCFAs), endogenous signals with important roles in lipid homeostasis, immunoregulation, maintenance of tissue barrier function (intestinal wall integrity) and regulation of systemic levels of inflammation [19, 20]. Prioritizing the health of the gut microbiome, through wholefoods PB diets, may also offer a several advantages for high-intensity training athletes to improve and manage metabolic, immune, and gastrointestinal disorders [21, 22].
Potential mechanisms linking a diet high in PB foods to improved athletic performance are limited, but not absent [23-25]. Importantly, many reports show that PB eating patterns neither improve nor hinder physical performance [26, 27] or resistance training goals [28], which may bring comfort to those committed to a PB/vegan lifestyle for reasons beyond personal gain. For example, in a review of eight studies, no acute differences were reported between PB diets and omnivorous diets when measuring muscular power, muscular strength, anaerobic or aerobic performance [26]. Similarly, other studies suggest that PB diets do not compromise endurance performance [29] and may facilitate aerobic capacity in athletes [30]. In this study [30] comparing young athletes who were either omnivores or a mixture of vegetarians composed of lacto-ovo-vegetarians and vegans for at least 2 years, the authors reported higher VO2max levels in female vegetarians without any differences in the male vegetarian group when compared with omnivores. In the same study, strength as measured by peak torque using leg extensions was comparable between vegetarians and omnivores in both men and women, reinforcing the notion that performance can be maintained if one choose to exclude ASF [30]. More recent findings also reported that a PB diet was not detrimental to endurance and muscle strength, and even showed superior submaximal endurance in vegans compared with omnivores [25]. It also appears that muscle and strength development (discussed later) with resistance training is supported by plant-sourced proteins to the same degree as ASF [31-35], with even stronger support at ingestions rates of protein reaching ~1.6 g/kg/day [34].
To date, no robust data support athletic performance improvements when switching to a PB diet, however, the notion that diets high in plant foods may offer performance benefits appears to have some merit and plausibility on several fronts [24]. Studies of diets in omnivores compared to vegetarians and vegans have consistently shown that PB diets are associated with lower BMIs and body fat levels [36, 37]. This is likely due to the higher fiber content and lower caloric density of PB diets which may help to optimize body composition, a key performance goal in fitness and sport [38]. There is also a preponderance of evidence to show that dietary strategies which achieve high carbohydrate (CHO) availability (i.e., to support energy expenditure) are associated with enhanced exercise capacity and sports performance, especially in high intensity endurance activities [39]. High CHO availability (from plant-sourced foods) prior to and during exercise enhances CHO (glycogen) storage and utilization, characteristics that are deemed beneficial to performance, as CHO oxidation produces more adenosine triphosphate (ATP) per unit of oxygen than fat [40]. High CHO
PB diets foster effective maintenance of glycogen concentrations which provide the fuel substrate to support ongoing higher intensity training, sport performance and recovery efforts [41]. In addition, by reducing blood viscosity and improving vascular flexibility and endothelial function, PB diets may improve blood flow and therefore oxygenation in skeletal and cardiac tissues [24], to further improve cardiovascular function.
Phytochemicals refer to active substances in PB diets that may provide unique benefits to highly active individuals. Thousands of dietary phytochemicals found in, for example, fruits, vegetables, nuts and seeds, legumes, grains, and seed oils possess antioxidant and anti-inflammatory properties [23, 42]. Whole-food plant-focused diets usually contain high amounts of phenolic acids, flavonoids and carotenoids, which have strong antioxidant properties, and therefore remove the excess of active oxygen in the body and protect cells from damage [43]. This can lead to reduced exercise induced oxidative stress and inflammation, and thus, enhanced endurance performance, reduced muscle damage, improved immune function and more efficient recovery from training and competition [23]. The comparability of PB diets to those higher in ASF with regard to performance along with the well-established health, environmental and ethical benefits of PB diets, make it an appealing choice for many athletes [27]. Although athletes should not necessarily expect to experience health and performance improvements by eliminating animal derived proteins, the inclusion of more plant-foods generally improves overall nutritional quality of one’s diet. As always, the impact of dietary changes for each individual will vary widely depending on their current or baseline dietary pattern. Increasing intakes and variety of plant-foods while also choosing fish and poultry over red meat more often, such as seen in a plant-focused Mediterranean diet appears to offer consistent health benefits [44]. This may be the most practical approach or “first step” for many athletes. It is also important to note that sustainability of a dietary pattern does not always correlate with health. Vegan and mostly-PB diets generally have much lower environmental impacts than diets containing meat [44, 45], but a vegan consuming excessive amounts of refined CHOs, added sugars and ultra-processed foods could be at greater risk for increasing body fat levels and chronic diseases than an omnivore consuming meat and a variety of healthy PB foods [46]. Finally, as with omnivorous diets, there are certain nutrients in PB diets that merit extra planning and attention to optimize health and performance for athletes. Nutrients to consider when following a strictly or mostly PB diet are canvassed below.
Macronutrients
Dietary Protein and Muscle Protein Synthesis
Resistance exercise training (RET) in combination with dietary protein supplementation or increased protein intake is common practice, in athletes, recreational weight-lifters and active gym-goers, with the aim of enhancing muscle growth and strength. A single session of exercise stimulates muscle protein synthesis (MPS) rates and, to a lesser extent, rates of muscle protein breakdown (MPB) [47]. Although post-exercise protein ingestion is widely applied as a strategy to augment post exercise anabolic stimulation to facilitate the muscle’s adaptive response to RET, the importance of protein ingestion during this “anabolic window” has been overestimated [34, 48, 49]. It appears that RET alone exerts the greatest increase in post training mixed-MPS (mostly myofibrillar but also include, mitochondrial and sarcoplasmic proteins) in the order of ~65% from resting basal values [48]. This fact warrants emphasis when discussing the efficacy of plant proteins for the purposes of MPS, as adopting an appropriate and robust RET-program is of overriding significance, whereas protein source and timing, although important, is a secondary factor to consider when optimizing adaptations to training [48]. Ingestion of adequate protein over the 24-h post RET session at a daily protein intake of ~1.6 g/kg per day from animal or plant sources, separated into ~0.25 g/kg doses appear to be more influential on adaptive changes with RET than amounts or timing of post-exercise protein supplementation [34].
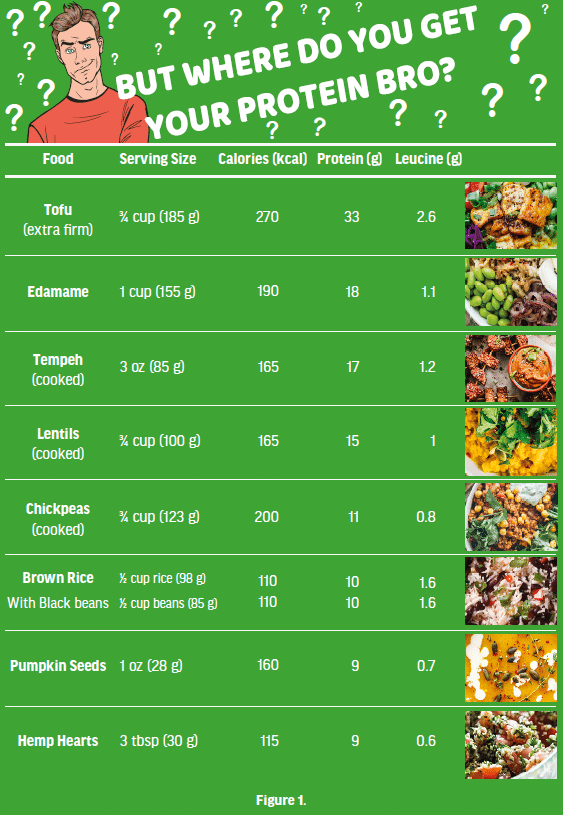
Historically, meat and other ASF and supplements (e.g., whey protein powders) have been viewed as key and critical components of an athlete’s diet. Many animal protein advocates question the adequacy of protein derived from a PB diet (Figure 1 & 2) to adequately support MPS for strength and hypertrophy, and recovery from high intensity exercise [50]. While PB protein sources often lack one or more amino acids (yet are usually adequate in 19 out of 20) in sufficient quantity to meet human nutritional needs [21], a combination of different proteins (Figure 2), including cereal-pulse combinations (e.g., rice and beans, chickpeas and quinoa), supplementation (plant protein powders such as soy, potato, rice and pea-based) and fortified food products (e.g., plant milks and PB meats), can help to overcome this in strict vegan or vegetarian diets [28]. The terms complete and incomplete are misleading in relation to plant protein. Ingesting protein from a variety of plant foods, over a 24- hour period, supplies enough of all indispensable (essential) amino acids when energy requirements are being met [51].
Robust trial evidence has shown leucine to be the dominant amino acid driving anabolism and skeletal muscle accretion, through MPS [52]. Whey protein has received much attention for muscle building due to its high level of leucine, whereas plant proteins tend to be lower in leucine and theoretically may not reach a similar anabolic threshold unless matched for leucine [33]. However, we generally do not generally eat single foods, we eat mixed meals that historically include a mix of plant and animal foods (e.g., chicken, rice, and vegetables). Mixed meals of solely plant-sourced foods shown in Figure 2 includes values for total protein and for the 9 essential amino acids. Three sample high plant protein meals are compared to the protein content of what are considered “very high quality” animal sourced proteins. In the given examples, protein and leucine content are equivalent to or surpass that which is contained in a serving of high-quality animal protein (a single protein food, no sides).
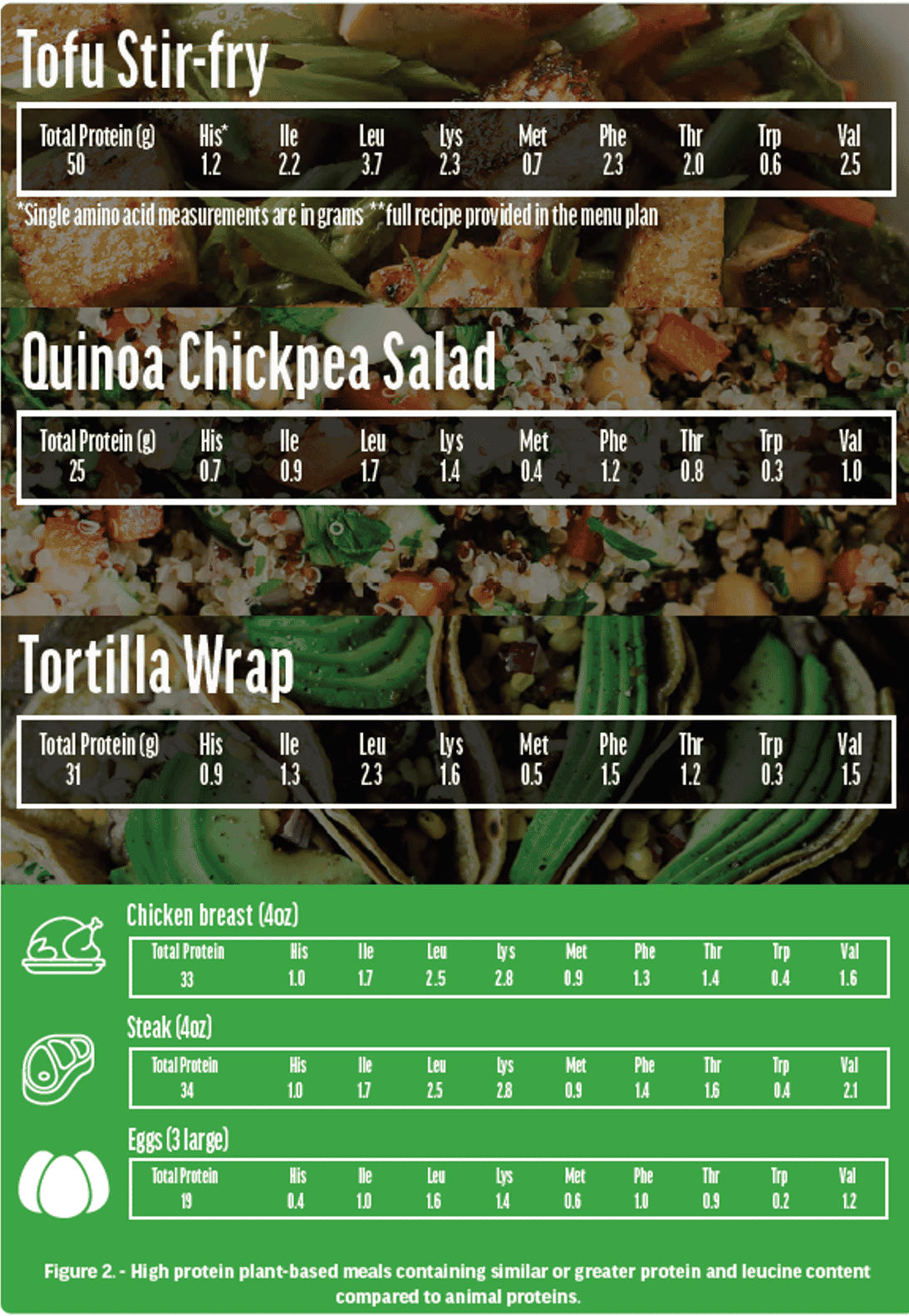
The amounts and proportions of amino acids consumed by vegetarians and vegans are typically more than sufficient to meet and exceed individual daily requirements, provided a reasonable variety of foods are consumed and energy intake needs are being met [32, 53] (Figure 2). Many studies have reported no differences in muscle strength of hypertrophy in response to RET and either plant vs animal sourced protein powder supplementation [31, 33, 35, 54]. One criticism of these comparisons however is that overall dietary (i.e., food protein in meals not protein supplements) protein intakes may or may not be restricted to PB sources (in the plant-protein supplemented group), therefore not eliminating the potential effects of animals-based proteins. To address this, in the most persuasive study to date, researchers compared the effects of a diet consisting of plant proteins exclusively vs an omnivorous diet on RT-induced muscle adaptations following a 12-week RT program in young males under an optimal protein intake (1.6 g/kg per day) combining whole foods plus supplemental protein (either soy or whey protein isolates). No significant differences were observed in increases in leg lean mass, muscle cross sectional area CSA (rectus femoris and vastus lateralis), fiber CSA (type I and type II muscle fiber), and muscle strength following diet and resistance training between the two groups, regardless of dietary protein source [28]. Although the beliefs and attitudes surrounding the superiority of animal-sourced proteins have dominated sports nutrition practices and guidelines for decades, a surge in plant protein research [28, 31, 35, 55] and the rampant growth of PB foods and food products in the marketplace [56] have sparked interest and wider acceptance of this dietary pattern.
But What About Quality of Plant Proteins?
Two requirements for a protein to be considered high quality, or complete, for humans are having adequate levels of indispensable (essential) amino acids to support human growth and development and being readily digested and absorbed. The Protein Digestibility Corrected Amino Acid Score (PDCAAS) was developed in 1989 to compare the indispensable amino acid content of a test protein (mg/g protein) to a theoretical reference protein thought to meet indispensable amino acid requirements (mg/g protein) for a given age group, creating a ratio known as the amino acid or chemical score [57]. The indispensable amino acid with the lowest ratio is referred to as the most limiting amino acid. Many plant-proteins are low in 1-2 indispensable amino acids, but this is easily rectified when a variety of plant-proteins are consumed throughout the day or at the same meal (see Figure 2). In 2011, the United Nations Food and Agriculture Organization (FAO) introduced an updated amino acid scoring system, the Digestible Indispensable Amino Acid Score (DIAAS) [58] to better reflect what amino acids are being digested as opposed to calculating values based on fecal nitrogen excretion. In general, most animal-based protein sources, such as milk, whey, casein, eggs, and beef, have excellent protein quality scores, as such, they are generally considered complete protein sources for supporting requirements for human growth and development. Similar to milk protein and whey, soy protein essentially has an equivalent value for bioavailability, and several more plant proteins (e.g., canola, potato, pea, barley, lentils and quinoa) are also considered to be of high quality when adequate amounts are ingested [59]. Although for some it may be difficult to take in relatively large amounts of protein from whole plant foods because they typically have a low percentage of protein, many PB meats, concentrated milks and an array of plant protein powder isolates and concentrates make it possible to consume 20 g or more of PB protein in one serving as ready-to-drink shakes, powder mixes, bars and other PB food products [55, 60]. In addition, as noted earlier, there does not appear to be a difference in the MPS response with subsequent strength and hypertrophic adaptations when protein intakes from a variety of sources (including plants only) reach ~1.6 g/kg per day, separated into ~0.25 g protein per kg per meal doses [28, 34].
Protein Recommendations
The Recommended Dietary Allowance (RDA) is 0.8 grams of protein per kilogram of body weight; however, the Position of the Academy of Nutrition and Dietetics, Dietitians of Canada, and the American College of Sports Medicine [61] suggests that athletes should consume 1.2-2.0 grams of protein per kg to support the body’s adaptations to exercise and increased protein turnover due to training. As with the RDA, no unique recommendation is given for athletes following vegetarian or vegan diets. As long as athletes consume adequate energy (kcal) from a well-balanced diet, vegetarian and vegan athletes should be able to consume the recommended amounts of protein through their diet [62]. While there is general agreement about the need for more protein, the debate about whether certain types of proteins are more conducive for building muscle and strength than others appears to be waning as high quality research into PB proteins and muscle and exercise physiology reveals that as long as higher protein intakes are being met (1.6 g/ kg per day) protein source appears to be irrelevant [28]. Studies examining the dietary intakes of vegan and vegetarian endurance athletes have also shown that these athletes are meeting their recommended daily protein [29, 30]. Food sources high in protein that are acceptable to sport-focused individuals following PB diets include soy products (tofu, tempeh, edamame, soymilk), pulses (beans, lentils, peas) and an array of plant-based meat substitutes and protein powders. Seeds, nuts, and whole grains, such as quinoa, and plant-based milks also contribute protein to the diet (see Table 1).
What About Soy?
Although foods made from soybeans such as tofu have been a staple in Asian diets for centuries, they have recently become popular in the West due to increased interests in PB diets and plant proteins. Research has consistently shown that soy protein significantly reduces LDL cholesterol by approximately 3-4% in adults [63, 64]. Soy can also alleviate hot flushes for some menopausal women. However, there have been some concerns that soy might lower a man’s testosterone levels. This is because the biologically active ingredients in soy – isoflavones – are phytoestrogens – plant based compounds that behave much like estrogens. Isoflavones were thought to act as endocrine or hormone disruptors, but this was only seen in animal studies in the 1990s [60].
Over the past three decades the health effects of soy foods and soybean isoflavones have been rigorously researched. Many human trials now show that adverse effects are also not seen on breast tissue or estrogen levels in women, or testosterone or estrogen levels, or sperm or semen parameters in men [60, 65]. The notion that soy foods can blunt testosterone has been debunked, although suspicion remains after rodent studies showed the possibility [65]. Accordingly, after extensive review, a recent metaanalysis provided the strongest evidence to date showing a lack of support for classifying isoflavones as endocrine disruptors [60]. Lastly, isoflavones are very low in soy protein isolate or concentrate (used in plant-based protein powders, veggies burgers, energy bars and other food products) because during processing as much as 90% of the isoflavone content is eliminated, compared to tofu or soymilk [60].
Several studies have compared whey, which is high in “anabolic leucine”, to soy protein. A recent metaanalysis of 9 clinical trials concluded that soy protein led to similar gains in muscle mass and strength as whey protein and other types of animal protein [31]. Similarly, a newly published study in older adults also support the efficacy of soy for maintaining and building lean mass and strength [54]. In summary, soy is a high-quality complete PB protein that has been shown to support lean body mass gains while also offering additional cardiometabolic advantages. Soy foods and supplements are recommended as part of a healthy well balance PB diet to support exercise and sport performance.
Omega-3 Fats
Omega-3 (n-3) polyunsaturated fatty acids (PUFA) are essential fatty acids with diverse biological effects that must be consumed through dietary or supplement sources due to insufficient PUFA synthesis in humans [66]. Some studies have shown that n-3 fats may influence the preservation of strength and enhance recovery from heavy exercise likely through their anti-inflammatory effects [67, 68], however other reports did not observe a beneficial effect of n-3 supplementation on appendicular lean mass, strength, physical performance or myofibrillar protein synthesis in older adults at risk of sarcopenia [69].
The n-3 fatty acid biosynthesis pathway converts ɑ-linolenic acid (ALA) to eicosapentaenoic (EPA) acid and further into docosahexaenoic acid (DHA, integral to the visual process and synaptic functioning). ALA is found mainly in nuts, seeds and plant oils, and DHA and EPA are found in fish and other seafood [70]. While intakes of plant n-3s or ALAs are generally higher in those following PB diets compared to omnivores, intakes of EPA and DHA are low in PB diets and virtually absent in vegans or strictly PB diets [70]. Despite the assumptions that PB eaters/vegans are at risk of deficiency due to low conversion of ALA to EPA and then to DHA [71], there does not appear to be evidence of deficiencies in adults [72]. Although fish-eaters in general have higher circulating levels of DHA, some reports have shown female vegans to potentially have higher (or at least not different) circulating DHA levels, with zero consumption, compared to fish-eaters [73], likely due to higher-than-average intakes of ALA. Recent evidence has brought into question aspects of the n-3 synthesis pathway that researchers thought were well understood, including the low rates of DHA synthesis (due to low conversion ALA to EPA to DHA) [74-76], highlighting the likelihood that PB eaters can maintain their serum DHA levels through consumption of ALAs only. Currently there is no recommended daily allowance (RDA) for EPA or DHA; therefore, those following a vegan or PB diet should focus on consuming adequate amounts of ALA (see Table 2 for ALA-rich foods) which may be higher than recommended by the current RDA [77]. Although DHA is unlikely unnecessary, vegans or those following a PB diet also have the option of obtaining DHA directly through vegan-friendly algal supplementation [77].
Micronutrients
Vitamin B12
Vitamin B12 (cobalamin) is not synthesized by humans and the only dietary sources are foods of animal origin and fortified foods (e.g., nutritional yeast flakes, breakfast cereals, energy bars, plant milks and others). Vitamin B12 is important for normal brain and nervous system functioning and helps to make DNA [78]. Symptoms of B12 deficiency include brain fog, memory problems, fatigue, lethargy, irritability, and moodiness. Vitamin B12 is also associated with red blood cell (RBC) formation and low levels may result in megaloblastic anemia [79]. Megaloblastic anemia limits the blood’s oxygen carrying capacity, thus reducing its availability to cells, which may negatively impact aerobic performance [79]. Individuals following PB diets must take particular care to consume enough vitamin B12, as it is mainly found in meat, eggs, and dairy products. Vitamin B12 deficiency is relatively common, especially among older people whether omnivore or PB eaters. Despite some reports in the popular media, studies on naturally occurring and Vitamin B12-containing plant-derived food sources show that only nori (seaweed), is suitable as a Vitamin B12 source for vegetarians [80, 81]. However, due to inconsistencies and variability in plant sources of B12 content, the only reliable PB/vegan sources of B12 are fortified breakfast cereals and nutritional yeasts, along with PB meat substitutes, PB milks and other PB fortified food products [82]. Those following a PB or vegan diet should take a B12 supplement (cyanocobalamin is recommended) at least once per week and track intakes derived from B12 fortified foods and beverages [62, 82].
Iron
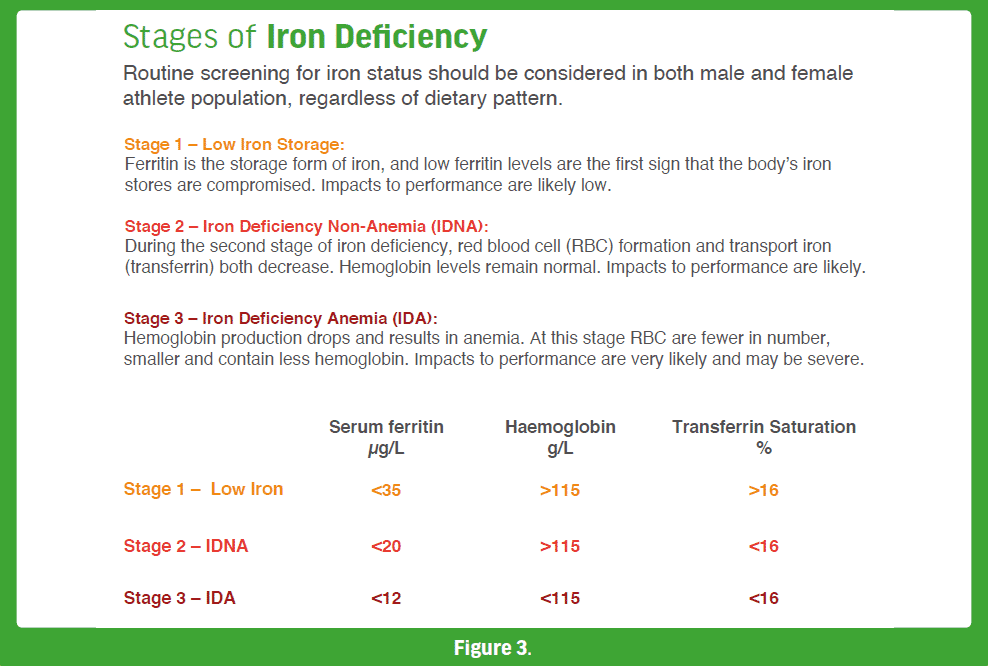
Iron is an essential mineral that plays a significant role in exercising individuals since it is required for oxygen transport and energy metabolism and building a strong immune system. Iron also enables erythropoiesis, the production of red blood cells (RBCs)[83]. RBCs supply oxygen to all organs and tissues in the body including skeletal muscle and the heart. Despite its importance, athlete populations, both omnivore and vegetarian, and especially females and endurance athletes, are commonly diagnosed with iron deficiency [84]. All types of iron deficiency may affect physical performance and should be treated accordingly through dietary
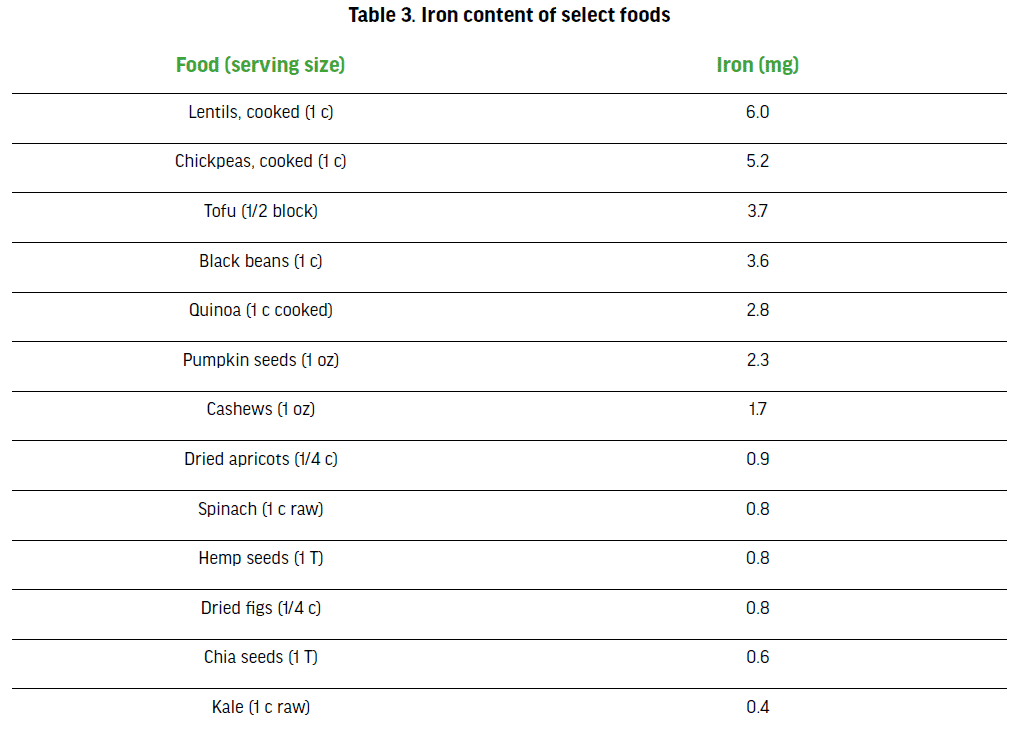
modifications (including fortified foods) or the use of supplements [85]. Low iron status is determined by measuring certain blood markers such as ferritin, hepcidin or transferrin. [84]. Figure 3 provides details on the three stages of iron deficiency with increasing impact on exercise and sport performance. Iron-deficiency anemia is the most common type of anemia among athletes, who have higher iron requirements due to increased erythropoietic drive through higher intensities and volumes of training as well as losses through sweat, foot strike hemolysis and GI bleeding [86, 87]. There are two major forms of iron found in food: heme iron, which is only found in animal products, and non-heme iron, which is found in both plant foods and animal products. Although the bioavailability of heme-iron is superior to nonheme iron, mineral absorption also depends on the iron status of the athlete (absorption is higher when iron stores are low) and dietary inhibitors, such as calcium, phytic acid, polyphenols and peptides (from partially digested proteins), and enhancers such as vitamin C and muscle tissue [88]. Owing to a diets rich in whole-grains, legumes (good iron sources) and the abundance of fortified mock meats and milk, both PB and vegans consume similar amounts of iron as omnivores [37, 89]; however, the risk of iron deficiency may [90] or may not [91] be greater due to lower bioavailability of iron from plant foods. Accordingly, recommended iron intakes for individuals following PB or vegan diets are 1.8 times higher than for omnivores who consume ASF [92]: 32 mg/day (vs. 18 mg/day) for premenopausal adult women and 14 mg/day (vs.8 mg/day) for adult men and postmenopausal women. Table 3 displays iron content of selected plant-based foods.
ZINC
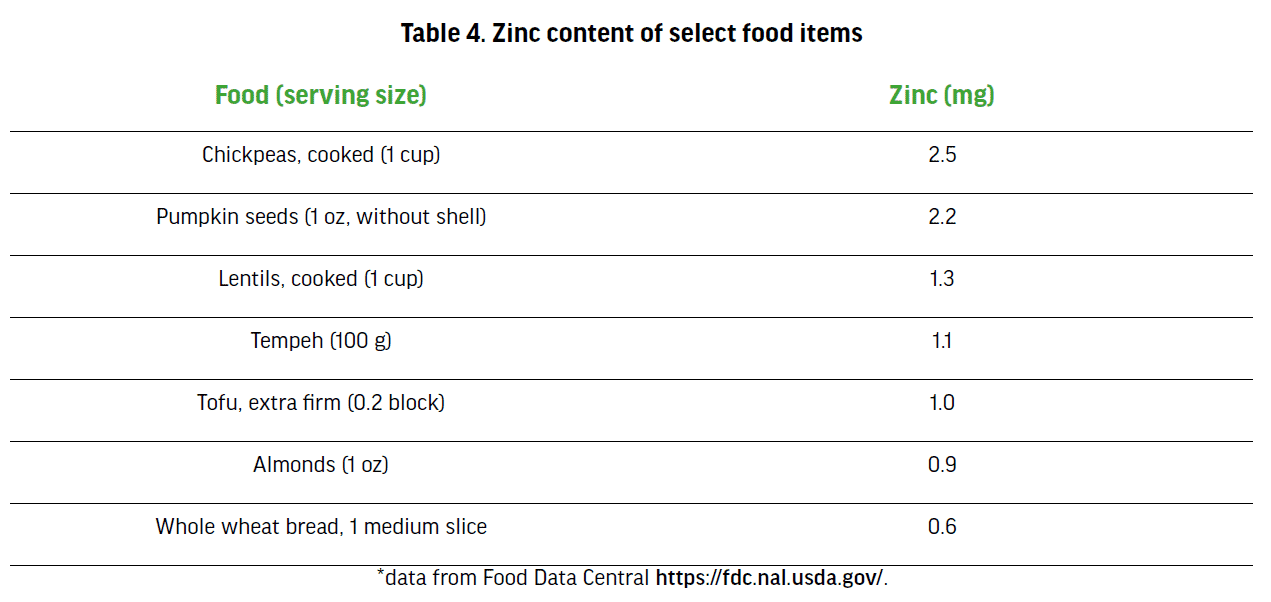
Zinc is a trace mineral abundantly distributed throughout all body tissues and fluids, and second only to iron among trace elements in the body. It is essential for multiple aspects of metabolism, including catalytic, structural and regulatory functions, and also plays an important role in the gene expression and the immune system [93]. Deficiency of this mineral can lead to hair loss, poor healing of wounds, immunological problems, skin problems and reproductive hormone imbalance[94]. Plant-based sources of zinc have lower bioavailability due to the presence of phytates (which are in legumes and whole grains) that bind zinc and inhibit its absorption [95]. Because of this reduced bioavailability, those following a vegetarian or strict PB diet may require up to 50% more zinc compared to omnivores [85]. This equates to male vegetarians needing 16.5 mg of zinc and female vegetarians needing 12 mg of zinc daily. Some preparation techniques that increase the bioavailability of plant sources of zinc include soaking and sprouting beans, nuts, seeds, and grains; leavening (bread has more bioavailable zinc compared to crackers); and using organic acids with zinc such as citric, malic, or lactic acid [62, 96]. Some researchers suggest supplementation may be advisable for vegan athletes [97], but this is not universally agreed upon. Soy, legumes, grains, seeds, nuts, beans, and fortified cereals are acceptable zinc sources for vegans and vegetarians. Table 4. provides a list of some zinc sources.
Calcium, Vitamin D, K2 and Bone Health
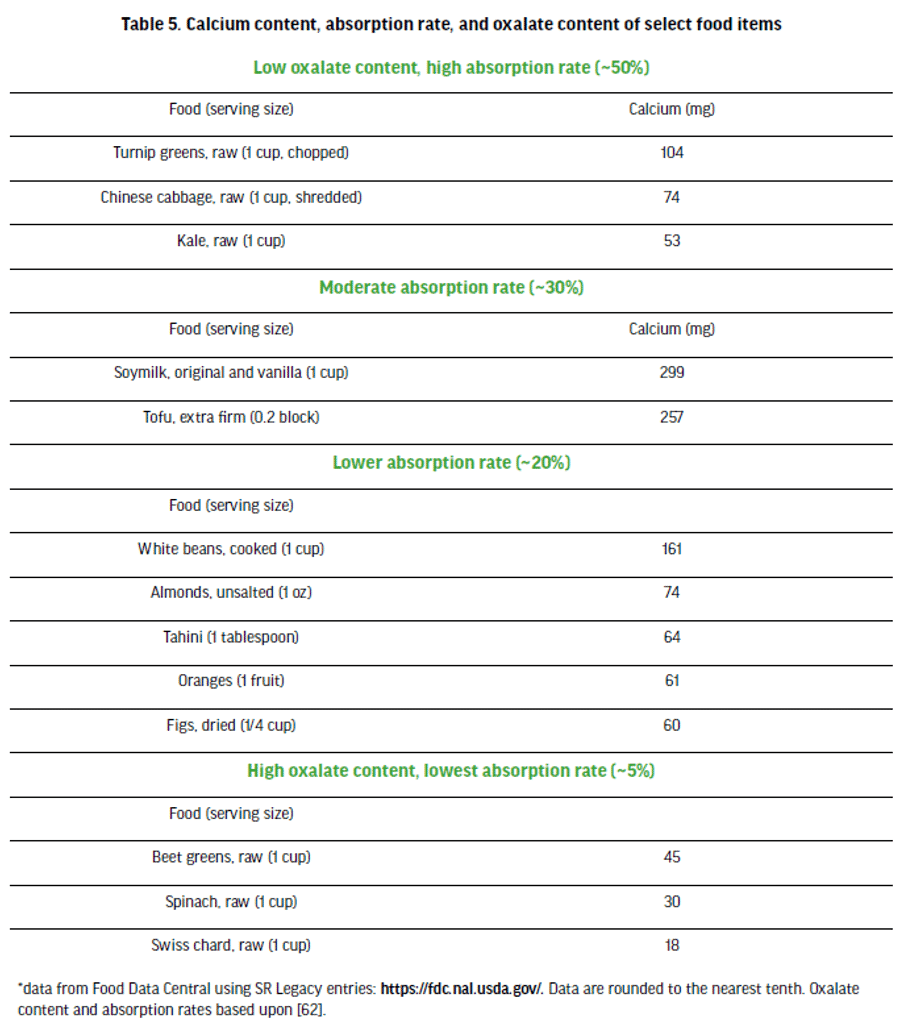
Calcium is an essential nutrient required for building and maintaining bone strength. It helps maintain the integrity of the skeleton as part of hydroxyapatite. Additionally, calcium is involved with muscle contraction, vasoconstriction and dilation, normal blood clotting, hormonal responses, and nerve conduction [98]. Inadequate dietary calcium and vitamin D increase the risk of low bone mineral density and stress fractures, which are a common injury in many sport disciplines [99]. The RDA for calcium in males and females ages 19-50 is 1,000 mg per day [98] although this can vary between countries. Vegetarians who include dairy in their diets tend to consume similar amounts or more calcium compared to omnivores, although people who follow vegan diets tend to consume less calcium [37, 89, 100]. Consuming a supplement of both vitamin D and calcium has been shown to be protective against bone fractures in vegans [101]. Plant sources of calcium may be less bioavailable due to the presence of oxalic acid [98] and to a lesser extent, phytates and fiber [62]. The International Olympic Committee consensus statement on dietary supplements and the high performance athlete posits that calcium and vitamin D are nutrients that often need to be supplemented for athletes, regardless of their dietary pattern [102]. Table 5 presents calcium content and variability in absorption rate of select plant-sourced calcium foods.
Vitamin D, along with calcium, is an important nutrient for optimizing bone health. Sufficient vitamin D is needed to absorb calcium and phosphate, contributes to normal functioning of most cell types in the body and it also plays an important role in immunity [103]. Vitamin D deficiency is associated with muscle weakness, fatigue, depression, and bone pain [103]. Currently the RDA for males and females ages 19-50 is 600 International Units (IU) per day [98]. The International Olympic Committee consensus statement on dietary supplements and the high-performance athlete suggests that vitamin D supplements are commonly required for athletes (not just for vegetarians) [102]. Vitamin D can be synthesized by the skin from UV light [104] or can be obtained through the diet, although it is not found naturally in many foods. Factors affecting how much vitamin D is synthesized by the skin’s exposure to sunlight include skin pigmentation, and amount and intensity of sun exposure, which is affected by the time of day, season, and latitude [105]. Research has shown no significant differences between vegetarians and omnivores with respect to serum 25-hydroxyvitamin D status [106]. Food sources providing vitamin D include UV-irradiated mushrooms and fortified foods such as plant based milks, orange juice, and cereals. It has been suggested that it may be beneficial for athletes to supplement 1,000-2,000 IUs/day, particularly if the athletes have minimal sun exposure [104].
Vitamin K is a family of essential, fat-soluble vitamins required for blood coagulation, but also involved in deposition and removal of calcium in various tissues. The family comprises two naturally active vitamers: vitamin K1 (phylloquinone) occurs in green vegetable and plant oils, and vitamin K2 (menaquinone). Menaquinones are principally of bacterial origin, but a small amount of MK-4 can be formed in mammals (including humans) through conversion of phylloquinone in plant feed or provided to animals as a feed additive [107]. Research shows that vitamin MK-4 derived from plant-sourced vitamin K1 is likely satisfactory to meet Vitamin K needs and animal-derived MK-4 sources are not necessary [108]. In summary, although data suggest that a well-planned, health-conscious lacto-ovo-vegetarian and vegan diet, including supplements, can meet athlete’s requirements for micronutrients it may still be prudent to periodically check blood values of primarily vitamin B12, vitamin D, and iron [109] as discussed below.
Taurine
Taurine is a sulfur-containing amino acid found in meat and fish, that plays many important physiological roles but is not an essential nutrient for healthy non-pregnant individuals following a PB/vegan diet [110]. Individuals following a strictly PB diet lack pre-formed dietary taurine due to its non-existence in plant foods, however, the in vivo synthesis of taurine using dietary methionine and cysteine precursors largely accommodates for the lack of dietary intake [111]. Methionine and cysteine are considered to be the principal sulfur-containing amino acids because they are two of the 20 canonical amino acids that are incorporated into proteins for functional and structural purposes in our body. Previous research has reported that plasma taurine concentrations were insignificantly and only slightly lower in vegans, compared to meat-eating omnivores, despite a large disparity in dietary intake [112]. Those following a PB diet should ensure adequate intakes of the taurine precursors methionine and cysteine, found in plant proteins such as nuts and seeds, and beans and lentils, respectively.
Should vegans test for nutritional deficiencies?
There are numerous advantages to a PB/vegan but unfortunately there can still be some vulnerabilities when it comes to ingesting adequate levels of certain nutrients. Blood tests are a good way of confirming optimal internal organ function and screening for potential vitamin and mineral deficiencies.
A Blood Profile for Vegans may include:
- Complete or Full Blood Count (FBC/CBC) provides detailed information about your red and white blood cells and can be used to detect conditions such as anaemias caused by iron and/or vitamin-B12 deficiency.
- Vitamin-B12 – a measurement of vitamin-B12 in your blood.
- Iron and Ferritin – a measurement of iron and ferritin (a marker of the body’s iron stores) levels (see Figure 3).
- Zinc deficiency can be diagnosed using a blood test, urine test, or hair analysis.
- 25(OH)D (Vitamin D) – is the form of vitamin D commonly measured to assess and monitor vitamin D status.
- Lipid Profile – this test measures triglycerides and levels of cholesterol and the sub-fractions including HDL cholesterol, and LDL cholesterol.
- HbA1C - measuring glycated haemoglobin (HbA1c) can provide an overall picture of what average blood sugar levels have been over a period of weeks/months.
If the results show that you are indeed deficient in some nutrients, you can make different dietary decisions by choosing more nutrient-specific and nutrient-dense foods. Changing your dietary goals may also require you to use supplements. Common supplements taken by vegans include omega-3s, iron, vitamin B12 and D3, and possibly iodine. A multivitamin/ mineral may also be warranted. Athletes should discuss the use of supplements with their health care provider or nutrition professional and ensure all supplements are batch tested to minimise risks from an anti-doping perspective.
Ergogenic Aids
Creatine
Increased muscle creatine content can help athletes generate quick, explosive movements at high intensities, enhance performance in strength and power events, and help to increase muscle mass by supporting increased volumes of resistance exercise [113]. Dietary intake of creatine is low or absent in individuals following a PB or vegan diet [114]. It was assumed that PB eaters who exclude dietary creatine sources and have lower muscle creatine storage [115] would experience greater ergogenic benefits. However, a recent review [114] found that creatine supplementation increased various parameters of performance in vegetarians and omnivores equally. Furthermore, the amount of creatine that is used in supplemental form to improve performance is 5 g per day as a maintenance dose, and 20 g per day for ~7 days as a loading dose, which cannot be practically achieved through dietary intakes alone. For example, an individual would have to consume 35 oz (992g) of beef, salmon or pork per day to reach the recommended daily maintenance dose of creatine for ergogenic goals of 5 g per day [114]. Emerging evidence also suggests that creatine supplementation may help to improve cognitive processing, especially in conditions characterized by brain creatine deficits, which could be induced by acute stressors (e.g., exercise, sleep deprivation, anxiety) or chronic, pathologic conditions (e.g., mild traumatic brain injury, aging, depression)[116]. Since creatine is deemed to be safe and appears to have both ergogenic and health benefits, the PB or vegan athlete may wish to ingest an ongoing maintenance dose of 1-3 g creatine supplements per day, to match the intakes of their omnivorous counterparts. See Nutrition X-change number 6 for more information on creatine.
Beta-Alanine
High-intensity exercise leads to a reduction in muscle pH (or increased acidity) due to hydrogen ion accumulation. Acidosis negatively impacts performance via a range of mechanisms, including reducing the capacity for muscle contraction and slowing the rate of ATP regeneration. Carnosine is a dipeptide formed by two amino acids, beta-alanine and L-histidine, and plays an important role in protecting against pH changes in skeletal muscle during exercise. Carnosine is found in skeletal muscle and the central nervous system, and is synthesised in situ from its rate-limiting precursor β-alanine [117]. Beta-alanine supplementation increases skeletal muscle carnosine content, and with it, the buffering capacity of the muscle (i.e. its capacity to regulate pH). This buffering ability has been shown to improves exercise performance, with its greatest effect in sustained, high-intensity efforts lasting between 30 seconds and 10 minutes. Muscle carnosine also tends to be lower in vegetarians compared to omnivores [118]. Although meat and poultry are the main sources of β-alanine in the diet there does not appear to be a significant relationship between dietary β-alanine consumption and muscle carnosine content [118]. Therefore, athletes following any dietary pattern would benefit from β-alanine supplementation to increase muscle carnosine concentrations [119]. Similar to creatine, the amount of β-alanine that is used in supplemental form to improve performance is 1.6 to 6.4 g per day for several days prior to an event, which is equivalent to approximately 24 oz (680g) of poultry per day to reach the equivalent β-alanine dose required to enhance performance [118]. Therefore, if applicable to the athlete both creatine and beta-alanine supplementation is recommended, rather than reliance on large portions of animals muscle for several days to elicit an ergogenic advantage. See Nutrition X-change number 9 for more information on beta alanine.
Summary
In summary, a well-designed whole foods PB diet that includes some fortified foods and optional supplements, can provide adequate macro- and micro-nutrient intakes in athletes and active individuals to support health and performance. Abundant choices in the marketplace also offer convenience and novelty with a wide selection of fortified food products and supplements for those choosing to follow a PB diet. Athletes in general should be taking extra care to plan out their sport nutrition strategies whether they choose to follow a strict an omnivore diet or a PB diet.
Although we have a vast amount of evidence in support of the nutritional adequacy and health benefits of PB diets, the sporting world’s perception of PB diets may still create challenges to widespread implementation. Sports nutrition guidelines are regularly reviewed to incorporate new scientific evidence, and it is now time to welcome the viability of PB options to athletes. With the increasing evidence that plant-based diets support optimal health, performance, recovery and body composition goals, the dissemination of accurate information to athletes and coaches is an important step in supporting the widespread transition to diets richer in traditional plants such as fruits, vegetables and whole grains as well as plant-proteins such as nuts, seeds, legumes and novel minimally processed meat and dairy alternatives.
Overview of the major food categories in a plant-based diet, with examples:
- Fruits: any type of fresh fruit including berries, apples, bananas, grapes, strawberries, citrus fruits, avocado etc. Dried fruits including figs dates, raisins, apricots and others.
- Vegetables: plenty of veggies including peppers, broccoli, corn, asparagus, avocados, lettuce, spinach, kale, peas, collards, and others.
- Tubers: root vegetables like potatoes, carrots, parsnips, sweet potatoes, beets, and others.
- Whole grains: grains, cereals, popcorn and other starches in their whole form, such as quinoa, brown rice, millet, whole wheat, oats, barley, and others.
- Nuts, Seeds & Oils: walnuts, almonds, cashews, brazil nuts, peanuts, and pumpkin, hemp, flax and chia seeds. Olive, pumpkinseed, sunflower, soybean, canola, avocado and sesame oil and others.
- Legumes: beans of any kind, lentils, pulses, and soy.
What does a Plant-Based Performance Diet Look Like?
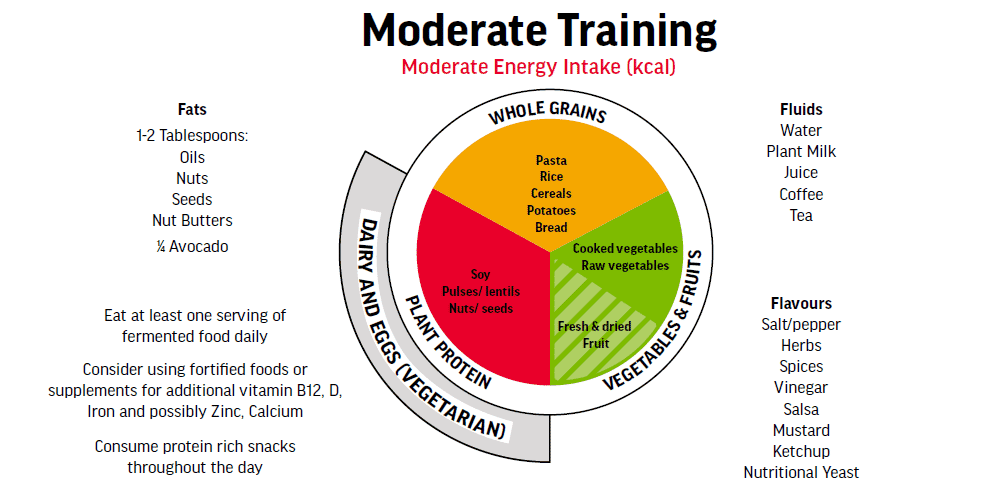
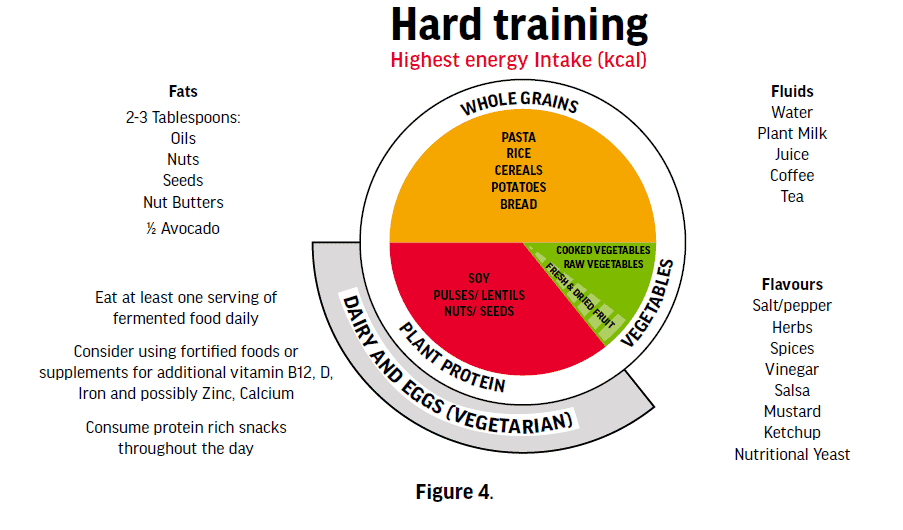
The Plant-Based Athlete’s Plate in Figure 4 is adapted from the original ‘The Athlete’s Plate®’ which is a visual tool designed to help sports dietitians working with athletes and athletes themselves adjust their dietary strategies to variable training loads and energy
expenditures during training and competition. The new Vegan/Vegetarian Athlete’s Plate® is based on the Athlete’s Plate® and was developed by University of Colorado, Colorado Springs Sport Nutrition Graduate Program in collaboration with the US Olympic Committee’s Food and Nutrition Services. The plates are now being scientifically tested (validated) against sport nutrition recommendations and will likely be published in 2022. This figure has modified some of the details according to filed experience with athletes, but you can find the original figure here: https://swell.uccs.edu/vegetarianvegan-athletes-plate.
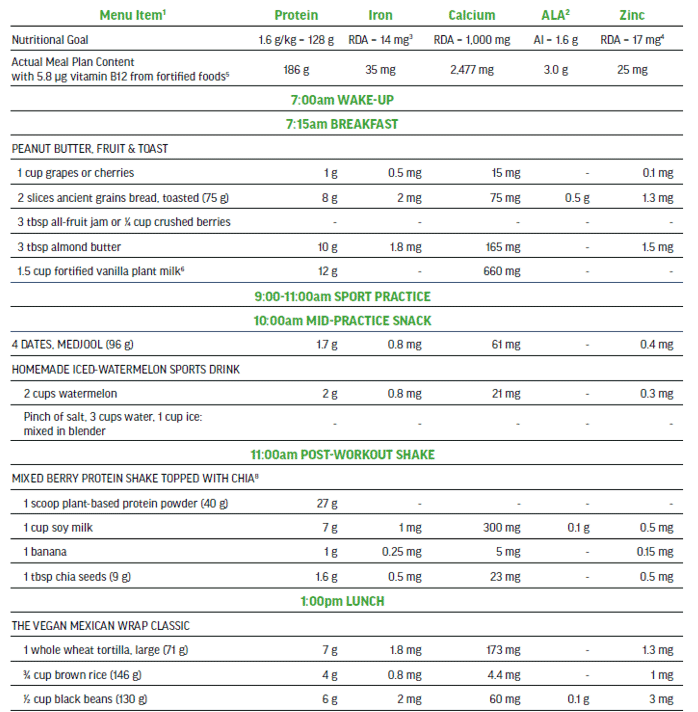
The 100% plant-based vegan menu in Figure 5 is a viable option to meet the needs of a high performance athlete that is seeking a highly palatable nutritionally-balanced sport-focused meal plan. This detailed three-day meal plan (Figure 5) provides variety while exceeding all of the macro and micro-nutrient requirements of an adult male training and competing for up to 15 hrs per week. The meal plan also supports maintenance of muscle mass to include recovery and repair. The nutritionally complete plan places emphasis on those nutrients in need of greater consideration when excluding animal-sourced foods from the diet.
Three-day Meal Plan for Adult Male Vegan Team Sport Athlete (80 kg).
Nutritional Content (across 3 days) for plant-sourced ‘nutrients of concern’: 613 g protein, 124 mg iron (non-heme), 7000 mg calcium, 18 g ALA, 75 mg zinc, and 18.7 μg vitamin B12.
Sample Menu Day 1: adult male training 12-15 hours per week, muscle mass maintenance.
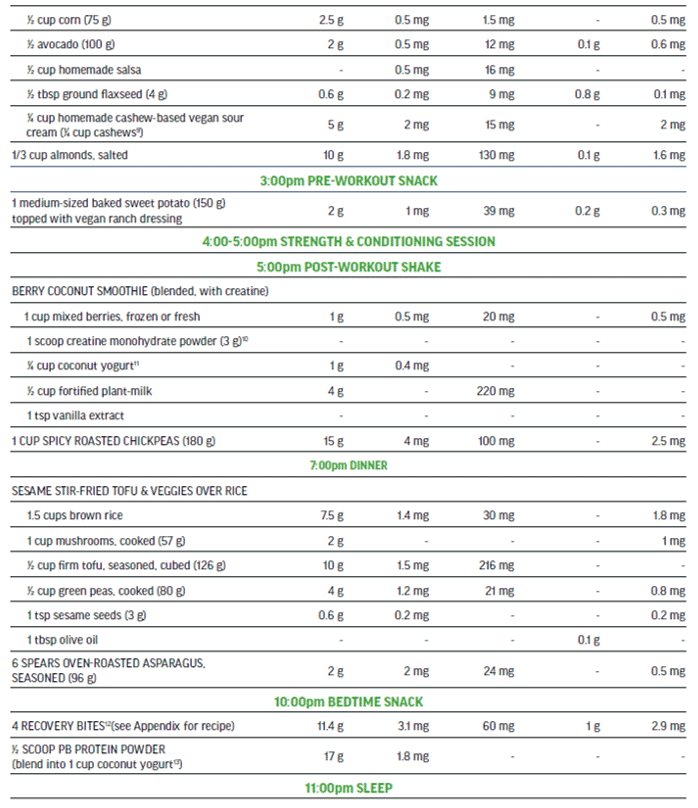
Sample Menu Day 2: adult male training 12-15 hours per week, muscle mass maintenance.
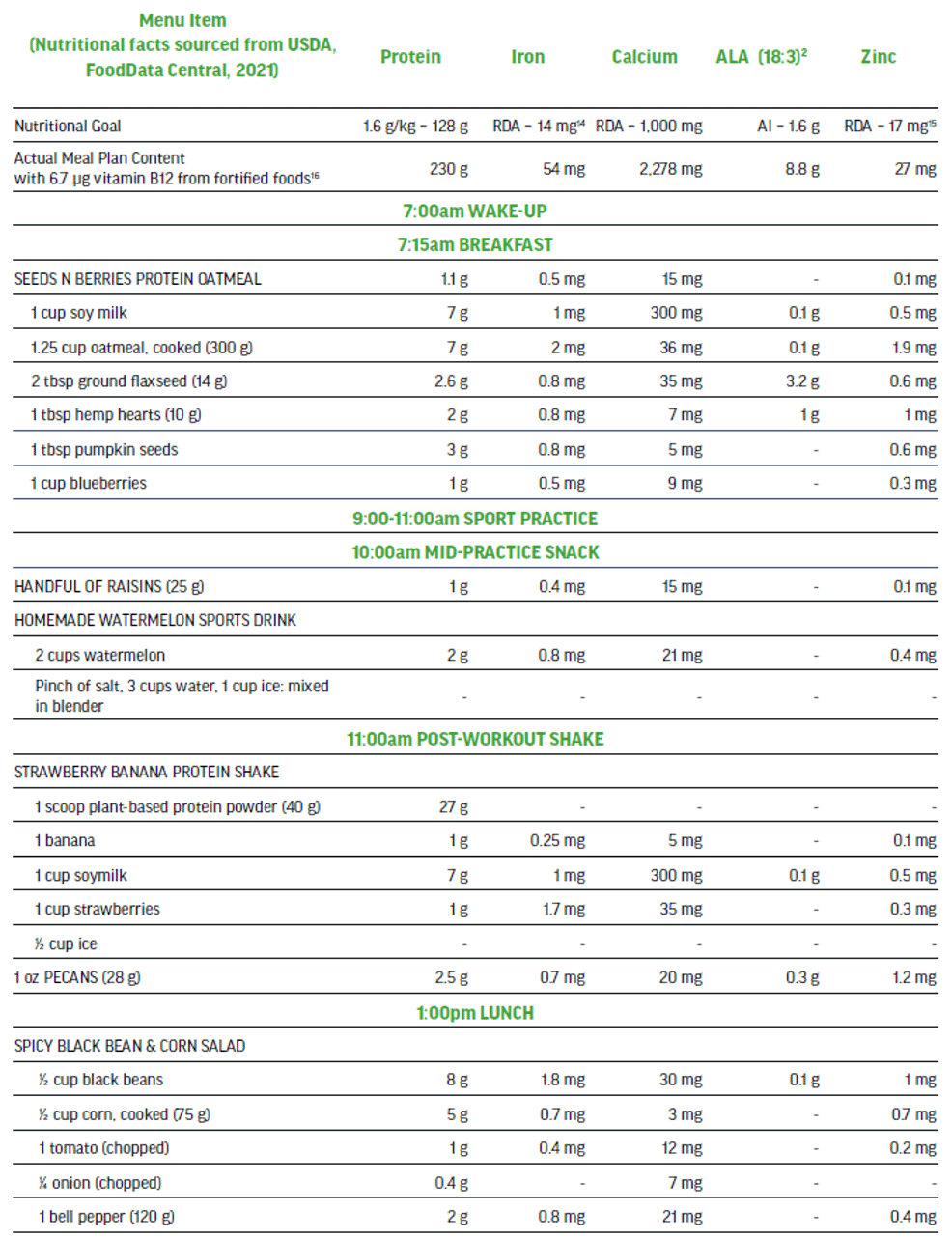
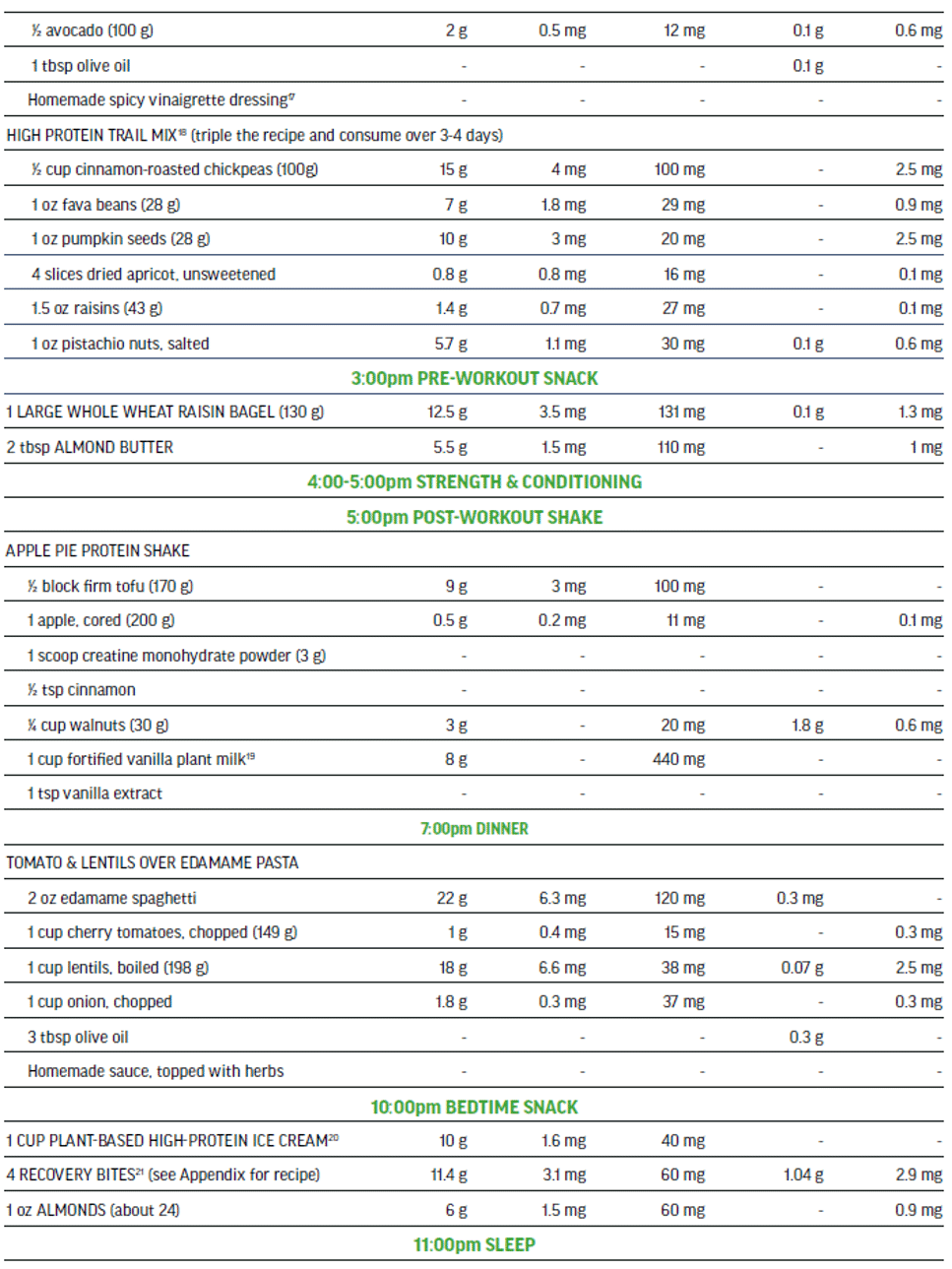
Sample Menu Day 3: adult male training 12-15 hours per week, muscle mass maintenance.
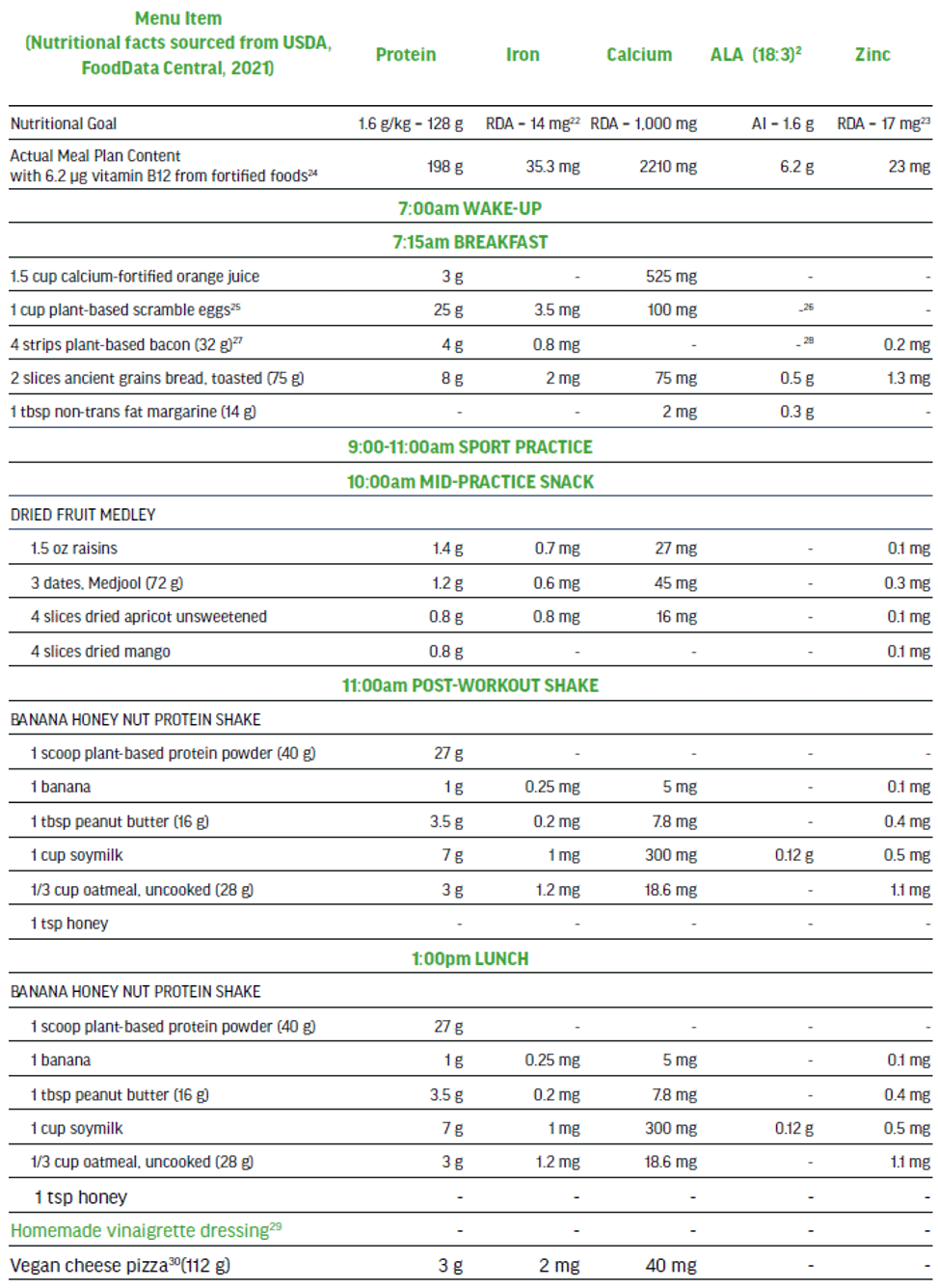
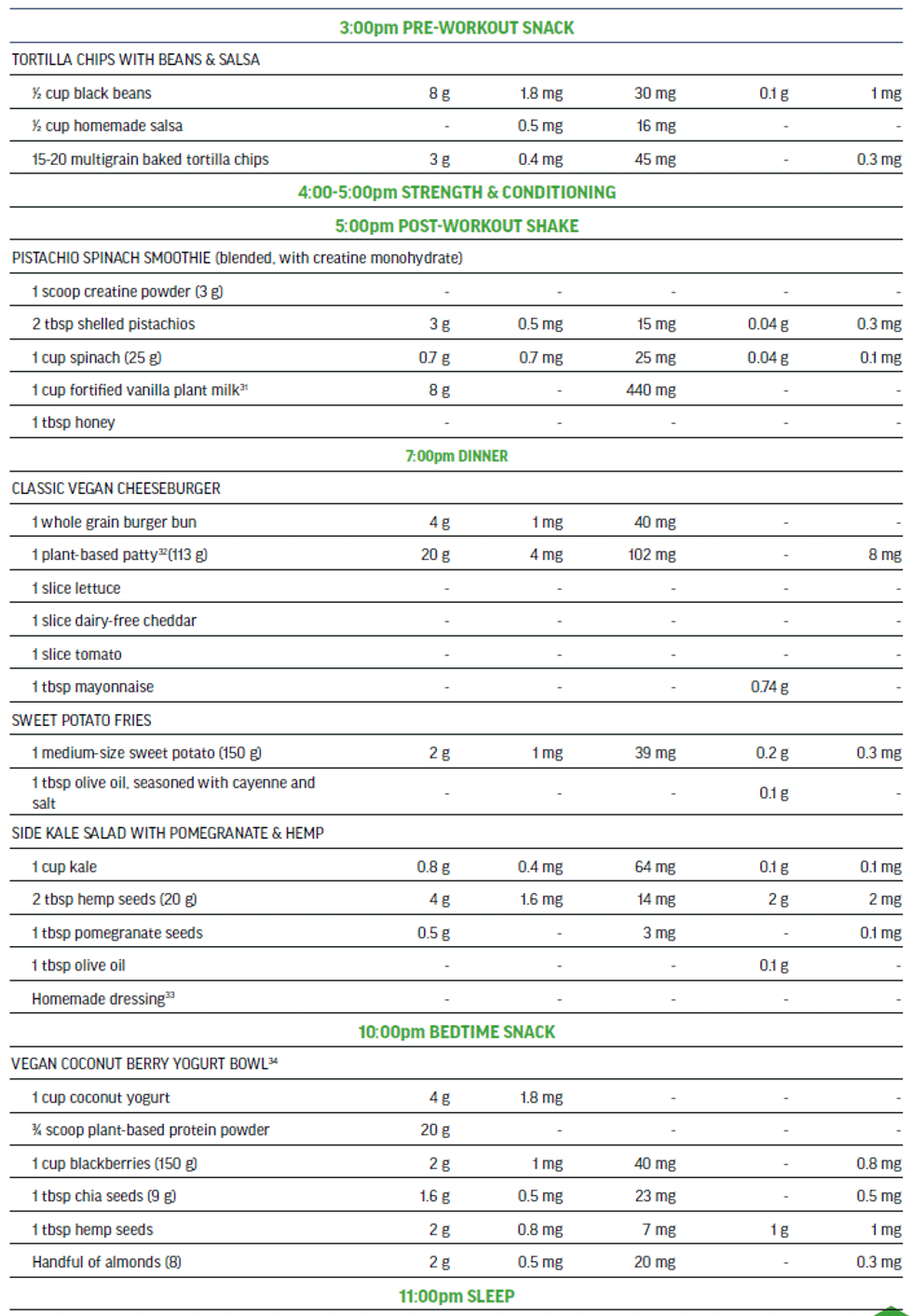
- Nutritional facts sourced from USDA, Food Data Central, 2021.
- Alpha-linolenic acid.
- The recommended dietary allowance (RDA) for iron is 1.8 times higher for vegans and vegetarians than for omnivores.
- The RDA for zinc is 1.5 times higher for vegans and vegetarians than for omnivores.
- Fortified vanilla plant milk and soymilk (RDA for vitamin B12: 2.4 μg/day).
- Based on Ripple Pea-Based Milk Vanilla.
- It is recommended that athletes invest in a good-quality blender for multi-ingredient protein shakes and smoothies.
- If no blender and on-the-go, mix protein powder with chilled plant milk and instead add chia seeds to today’s lunch wrap.
- Also requires water, lemon juice, apple cider vinegar, sea salt, and Dijon mustard.
- If applicable to athlete’s sport, 0.33 scoop (1.5 g) of beta-alanine powder is also recommended.
- Based on Co Yo Probiotic Coconut Milk Yogurt, available in US, UK, and Australia.
- Sunflower seeds, pumpkin seeds, oats, dates, goji berries, hemp seeds.
- Based on Co Yo Probiotic Coconut Milk Yogurt, available in US, UK, and Australia.
- The RDA for iron is 1.8 times higher for vegans and vegetarians than for omnivores.
- The RDA for zinc is 1.5 times higher for vegans and vegetarians than for omnivores.
- Soymilk and fortified vanilla plant milk.
- Also requires vinegar, Dijon mustard, maple syrup, garlic, and salt and pepper.
- Athletes are encouraged to alternate between different types of nuts, seeds, and dried fruits that are low in added sugars and added fats.
- Based on Ripple Pea-Based Milk Vanilla.
- Based on Snow Monkey Dairy-Free Anytime Desserts.
- Sunflower seeds, pumpkin seeds, oats, dates, goji berries, hemp seeds.
- The RDA for iron is 1.8 times higher for vegans and vegetarians than for omnivores.
- The RDA for zinc is 1.5 times higher for vegans and vegetarians than for omnivores
- Plant-based bacon, soymilk, and fortified vanilla plant milk
- Based on JUST Egg, Plant-Based Scramble
- Contains 7.5 grams of polyunsaturated fatty acids
- Based on Morning Star Farms Veggie Bacon Strips
- Contains 5 grams of polyunsaturated fatty acids
- Also requires vinegar, Dijon mustard, fresh lemon juice, maple syrup, garlic, and salt and pepper
- Based on Bold Organics Vegan Cheese Pizza
- Based on Ripple Pea-Based Milk Vanilla
- Based on Beyond Meat Beyond Burger Plant-Based Patties
- Requires lemon juice, garlic, salt
- Stir protein powder into yogurt, then top with berries, seeds, and crushed almonds
- Based on Co Yo Probiotic Coconut Milk Yogurt, available in US, UK, and Australia
Appendix
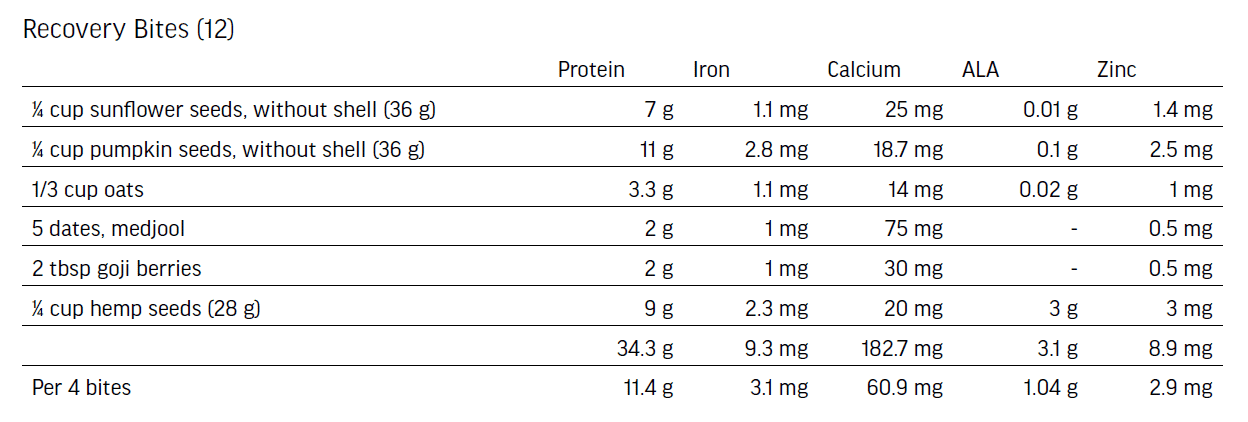 Nutritional Boost Protein Add-Ons:
Nutritional Boost Protein Add-Ons:
- For an extra 6g protein & 20 mg calcium: 4 tbsp hummus & vegetables.
- For an extra 4g protein & 0.5 mg iron: handful of wasabi peas.
- For an extra 16g protein & 2.3 mg zinc: 1 cup of edamame.
- For an extra 11g protein: 1.5 oz (42 g) roasted fava beans and peas can be added to salads and trail-mixes.
- For an extra 11g protein: 1/3 cup (52 g) almonds can be added to salads and trail-mixes.
- For an extra 20g protein, 3.8 mg iron, & 735 mg calcium: 1.5 cup high-protein plant-based drink (based on Original Silk® Ultra Plant-Based Protein Drink).
References
- Esselstyn, C.B., A plant-based diet and coronary artery disease: a mandate for effective therapy. J Geriatr Cardiol, 2017. 14(5): p. 317-320.
- Hemler, E.C. and F.B. Hu, Plant-Based Diets for Personal, Population, and Planetary Health. Adv Nutr, 2019. 10(Suppl_4): p. S275-S283.
- Panizza, S., If Veganism Is Not a Choice: The Moral Psychology of Possibilities in Animal Ethics. Animals (Basel), 2020. 10(1).
- Heianza, Y., et al., Healthful plantbased dietary patterns, genetic risk of obesity, and cardiovascular risk in the UK biobank study. Clin Nutr, 2021. 40(7): p. 4694-4701.
- Kahleova, H., S. Levin, and N.D. Barnard, Vegetarian Dietary Patterns and Cardiovascular Disease. Prog Cardiovasc Dis, 2018. 61(1): p. 54-61.
- Lee, K.W., et al., Effects of Vegetarian Diets on Blood Pressure Lowering: A Systematic Review with Meta-Analysis and Trial Sequential Analysis. Nutrients, 2020. 12(6)
- Kahleova, H., et al., A Plant-Based Dietary Intervention Improves Beta-Cell Function and Insulin Resistance in Overweight Adults: A 16-Week Randomized Clinical Trial. Nutrients, 2018. 10(2).
- Viguiliouk, E., et al., Effect of vegetarian dietary patterns on cardiometabolic risk factors in diabetes: A systematic review and meta-analysis of randomized controlled trials. Clin Nutr, 2019. 38(3): p.1133-1145.
- Li, S.S., et al., Effect of Plant Protein on Blood Lipids: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc, 2017. 6(12).
- Catsburg, C., et al., Dietary patterns and breast cancer risk: a study in 2 cohorts. Am J Clin Nutr, 2015. 101(4): p. 817-23.
- Tantamango-Bartley, Y., et al., Are strict vegetarians protected against prostate cancer? Am J Clin Nutr, 2016. 103(1): p. 153-60.
- Orlich, M.J., et al., Vegetarian dietary patterns and the risk of colorectal cancers. JAMA Intern Med, 2015. 175(5): p. 767-76.
- Papier, K., et al., Comparison of Major Protein-Source Foods and Other Food Groups in Meat-Eaters and Non-Meat-Eaters in the EPIC-Oxford Cohort. Nutrients, 2019. 11(4).
- Noh, H., et al., Taxonomic Composition and Diversity of the Gut Microbiota in Relation to Habitual Dietary Intake in Korean Adults. Nutrients, 2021. 13(2).
- Glick-Bauer, M. and M.C. Yeh, The health advantage of a vegan diet: exploring the gut microbiota connection. Nutrients, 2014. 6(11): p. 4822-38.
- Davani-Davari, D., et al., Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods, 2019. 8(3).
- Cai, J., et al., High animal protein diet and gut microbiota in human health. Crit Rev Food Sci Nutr, 2021: p. 1-13.
- Sakkas, H., et al., Nutritional Status and the Influence of the Vegan Diet on the Gut Microbiota and Human Health. Medicina (Kaunas), 2020. 56(2).
- Hills, R.D., Jr., et al., Gut Microbiome: Profound Implications for Diet and Disease. Nutrients, 2019. 11(7).
- Valdes, A.M., et al., Role of the gut microbiota in nutrition and health. BMJ, 2018. 361: p. k2179.
- Crowson, M.M. and S.A. McClave, Does the Intestinal Microbiome Impact Athletic Performance? Curr Gastroenterol Rep, 2020. 22(11): p. 53.
- Milajerdi, A., et al., Association of Dietary Fiber, Fruit, and Vegetable Consumption with Risk of Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Adv Nutr, 2021. 12(3): p. 735-743.
- Trapp, D., W. Knez, and W. Sinclair, Could a vegetarian diet reduce exercise-induced oxidative stress? A review of the literature. J Sports Sci, 2010. 28(12): p. 1261-8.
- Barnard, N.D., et al., Plant-Based Diets for Cardiovascular Safety and Performance in EnduranceSports. Nutrients, 2019. 11(1).
- Boutros, G.H., et al., Is a vegan diet detrimental to endurance and muscle strength? Eur J Clin Nutr, 2020.
- Craddock, J.C., Y.C. Probst, and G.E. Peoples, Vegetarian and Omnivorous Nutrition - Comparing Physical Performance. Int J Sport Nutr Exerc Metab, 2016. 26(3): p. 212-20.
- Meyer, N. and A. Reguant-Closa, “Eat as If You Could Save the Planet and Win!” Sustainability Integration into Nutrition for Exercise and Sport. Nutrients, 2017. 9(4).
- Hevia-Larrain, V., et al., High-Protein Plant-Based Diet Versus a Protein-Matched Omnivorous Diet to Support Resistance Training Adaptations: A Comparison Between Habitual Vegans and Omnivores. Sports Med, 2021. 51(6): p. 1317-1330.
- Nebl, J., et al., Characterization, dietary habits and nutritional intake of omnivorous, lacto-ovovegetarian and vegan runners–a pilot study. BMC nutrition, 2019. 5(1): p. 51.
- Lynch, H.M., C.M. Wharton, and C.S. Johnston, Cardiorespiratory fitness and peak torque differences between vegetarian and omnivore endurance athletes: A cross-sectional study. Nutrients, 2016. 8(11): p. 726.
- Messina, M., et al., No Difference Between the Effects of Supplementing With Soy Protein Versus Animal Protein on Gains in Muscle Mass and Strength in Response to Resistance Exercise. Int J Sport Nutr Exerc Metab, 2018. 28(6): p. 674-685.
- Lynch, H., C. Johnston, and C. Wharton, Plant-Based Diets: Considerations for Environmental Impact, Protein Quality, and Exercise Performance. Nutrients, 2018. 10(12).
- Lynch, H.M., et al., No Significant Differences in Muscle Growth and Strength Development When Consuming Soy and Whey Protein Supplements Matched for Leucine Following a 12 Week Resistance Training Program in Men and Women: A Randomized Trial. Int J Environ Res Public Health, 2020. 17(11).
- Morton, R.W., et al., A systematic review, metaanalysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br J Sports Med, 2018. 52(6): p. 376-384.
- Moon, J.M., et al., Effects of daily 24-gram doses of rice or whey protein on resistance training adaptations in trained males. J Int Soc Sports Nutr, 2020. 17(1): p. 60.
- Najjar, R.S. and R.G. Feresin, Plant-Based Diets in the Reduction of Body Fat: Physiological Effects and Biochemical Insights. Nutrients, 2019. 11(11).
- Rizzo, N.S., et al., Nutrient profiles of vegetarian and nonvegetarian dietary patterns. Journal of the Academy of Nutrition and Dietetics, 2013. 113(12): p. 1610-1619.
- Aragon, A.A., et al., International society of sports nutrition position stand: diets and body composition. J Int Soc Sports Nutr, 2017. 14: p. 16.
- Stellingwerff, T. and G.R. Cox, Systematic review: Carbohydrate supplementation on exercise performance or capacity of varying durations. Appl Physiol Nutr Metab, 2014. 39(9): p. 998-1011.
- Shaw, D.M., et al., Effect of a Ketogenic Diet on Submaximal Exercise Capacity and Efficiency in Runners. Med Sci Sports Exerc, 2019. 51(10): p. 2135-2146.
- Kanter, M., High-Quality Carbohydrates and Physical Performance: Expert Panel Report. Nutr Today, 2018. 53(1): p. 35-39.
- Upadhyay, S. and M. Dixit, Role of Polyphenols and Other Phytochemicals on Molecular Signaling. Oxid Med Cell Longev, 2015. 2015: p. 504253.
- Guan, R., et al., A review of dietary phytochemicals and their relation to oxidative stress and human diseases. Chemosphere, 2021. 271: p. 129499.
- Dernini, S. and E.M. Berry, Mediterranean Diet: From a Healthy Diet to a Sustainable Dietary Pattern. Front Nutr, 2015. 2: p. 15.
- Cacau, L.T., et al., Development and Validation of an Index Based on EAT-Lancet Recommendations: The Planetary Health Diet Index. Nutrients, 2021. 13(5).
- Gehring, J., et al., Consumption of Ultra-Processed Foods by Pesco-Vegetarians, Vegetarians, and Vegans: Associations with Duration and Age at Diet Initiation. J Nutr, 2021. 151(1): p. 120-131.
- Phillips, S.M., et al., Mixed muscle protein synthesis and breakdown after resistance exercise in humans. Am J Physiol, 1997. 273(1 Pt 1): p. E99-107.
- Reidy, P.T. and B.B. Rasmussen, Role of Ingested Amino Acids and Protein in the Promotion of Resistance Exercise-Induced Muscle Protein Anabolism. J Nutr, 2016. 146(2): p. 155-83.
- Schoenfeld, B.J., A.A. Aragon, and J.W. Krieger, The effect of protein timing on muscle strength and hypertrophy: a meta-analysis. J Int Soc Sports Nutr, 2013. 10(1): p. 53.
- van Vliet, S., N.A. Burd, and L.J. van Loon, The Skeletal Muscle Anabolic Response to Plant- versus Animal-Based Protein Consumption. J Nutr, 2015. 145(9): p. 1981-91.
- Mariotti, F. and C.D. Gardner, Dietary Protein and Amino Acids in Vegetarian Diets-A Review. Nutrients, 2019. 11(11).
- Phillips, S.M., The impact of protein quality on the promotion of resistance exercise-induced changes in muscle mass. Nutr Metab (Lond), 2016. 13: p. 64.
- Gardner, C.D., et al., Maximizing the intersection of human health and the health of the environment with regard to the amount and type of protein produced and consumed in the United States. Nutr Rev, 2019. 77(4): p. 197-215.
- Li, C., et al., Daily Supplementation With Whey, Soy, or Whey-Soy Blended Protein for 6 Months Maintained Lean Muscle Mass and Physical Performance in Older Adults With Low Lean Mass. J Acad Nutr Diet, 2021. 121(6): p. 1035-1048 e6.
- McClements, D.J. and L. Grossmann, The science of plant-based foods: Constructing nextgeneration meat, fish, milk, and egg analogs. Compr Rev Food Sci Food Saf, 2021. 20(4): p. 4049-4100.
- Institute, G.F., Plant-based foods in the U.S. are a $7 billion market. 2021.
- Eggum, B., Comments on report of a jointFAO/WHO expert consultation on protein quality evaluation, Rome 1990. Z Ernahrungswiss, 1991. 30(2): p. 81-8.
- Schaafsma, G., Advantages and limitations of the protein digestibility-corrected amino acid score (PDCAAS) as a method for evaluating protein quality in human diets. Br J Nutr, 2012. 108 Suppl 2: p. S333-6.
- Hertzler, S.R., et al., Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function. Nutrients, 2020. 12(12).
- Messina, M., et al., Neither soyfoods nor isoflavones warrant classification as endocrine disruptors: a technical review of the observational and clinical data. Crit Rev Food Sci Nutr, 2021: p. 1-57.
- Thomas, D.T., K.A. Erdman, and L.M. Burke, Position of the academy of nutrition and dietetics, dietitians of canada, and the american college of sports medicine: Nutrition and athletic performance. Journal of the Academy of Nutrition and Dietetics, 2016. 116(3): p. 501-528.
- Melina, V., W. Craig, and S. Levin, Position of the Academy of Nutrition and Dietetics: Vegetarian Diets. Journal of the Academy of Nutrition and Dietetics, 2016. 116(12): p. 1970-1980.
- Blanco Mejia, S., et al., A Meta-Analysis of 46 Studies Identified by the FDA Demonstrates that Soy Protein Decreases Circulating LDL and Total Cholesterol Concentrations in Adults. J Nutr, 2019. 149(6): p. 968-981.
- Jenkins, D.J.A., et al., Cumulative Meta-Analysis of the Soy Effect Over Time. J Am Heart Assoc, 2019. 8(13): p. e012458.
- Reed, K.E., et al., Neither soy nor isoflavone intake affects male reproductive hormones: An expanded and updated meta-analysis of clinical studies. Reprod Toxicol, 2021. 100: p. 60-67.
- Yashodhara, B.M., et al., Omega-3 fatty acids: a comprehensive review of their role in health and disease. Postgrad Med J, 2009. 85(1000): p. 84-90.
- Heileson, J.L. and L.K. Funderburk, The effect of fish oil supplementation on the promotion and preservation of lean body mass, strength, and recovery from physiological stress in young, healthy adults: a systematic review. Nutr Rev, 2020.
- Tsuchiya, Y., et al., 4-week eicosapentaenoic acid-rich fish oil supplementation partially protects muscular damage following eccentric contractions. J Int Soc Sports Nutr, 2021. 18(1): p. 18.
- Murphy, C.H., et al., Does supplementation with leucine-enriched protein alone and in combination with fish-oil-derived n-3 PUFA affect muscle mass, strength, physical performance, and muscle protein synthesis in well-nourished older adults? A randomized, double-blind, placebo-controlled trial. Am J Clin Nutr, 2021. 113(6): p. 1411-1427.
- Sanders, T.A., DHA status of vegetarians. Prostaglandins Leukot Essent Fatty Acids, 2009. 81(2-3): p. 137-41.
- Saunders, A.V., B.C. Davis, and M.L. Garg, Omega-3 polyunsaturated fatty acids and vegetarian diets. Med J Aust, 2013. 199(S4): p. S22-6.
- Metherel, A.H. and R.P. Bazinet, Updates to the n-3 polyunsaturated fatty acid biosynthesis pathway: DHA synthesis rates, tetracosahexaenoic acid and (minimal) retroconversion. Prog Lipid Res, 2019. 76: p. 101008.
- Welch, A.A., et al., Dietary intake and status of n-3 polyunsaturated fatty acids in a population of fish-eating and non-fish-eating meat-eaters, vegetarians, and vegans and the product-precursor ratio [corrected] of alpha-linolenic acid to long-chain n-3 polyunsaturated fatty acids: results from the EPIC-Norfolk cohort. Am J Clin Nutr, 2010. 92(5): p. 1040-51.
- Metherel, A.H., et al., Compound-specific isotope analysis reveals no retroconversion of DHA to EPA but substantial conversion of EPA to DHA following supplementation: a randomized control trial. Am J Clin Nutr, 2019. 110(4): p. 823-831.
- Domenichiello, A.F., A.P. Kitson, and R.P. Bazinet, Is docosahexaenoic acid synthesis from alpha-linolenic acid sufficient to supply the adult brain? Prog Lipid Res, 2015. 59: p. 54-66.
- Metherel, A.H., et al., Whole-body DHA synthesis-secretion kinetics from plasma eicosapentaenoic acid and alpha-linolenic acid in the free-living rat. Biochim Biophys Acta, 2016. 1861(9 Pt A): p. 997-1004.
- Burns-Whitmore, B., et al., Alpha-Linolenic and Linoleic Fatty Acids in the Vegan Diet: Do They Require Dietary Reference Intake/Adequate Intake Special Consideration? Nutrients, 2019. 11(10).
- Hooshmand, B., et al., Association of Vitamin B12, Folate, and Sulfur Amino Acids With Brain Magnetic Resonance Imaging Measures in Older Adults: A Longitudinal Population-Based Study. JAMA Psychiatry, 2016. 73(6): p. 606-13.
- Nagao, T. and M. Hirokawa, Diagnosis and treatment of macrocytic anemias in adults. J Gen Fam Med, 2017. 18(5): p. 200-204.
- Pawlak, R., S.E. Lester, and T. Babatunde, The prevalence of cobalamin deficiency among vegetarians assessed by serum vitamin B12: a review of literature. Eur J Clin Nutr, 2014. 68(5): p. 541-8.
- Watanabe, F., et al., Vitamin B(1)(2)-containing plant food sources for vegetarians. Nutrients, 2014. 6(5): p. 1861-73.
- Medicine, I.I.o. Vitamin B12 Fact Sheet for Health Professionals. 2020; Available from: https://ods.od.nih.gov/factsheets/VitaminB12- HealthProfessional/#en5.
- Anker, S.D., et al., Ferric carboxymaltose in patients with heart failure and iron deficiency. N Engl J Med, 2009. 361(25): p. 2436-48.
- Clenin, G., et al., Iron deficiency in sports - definition, influence on performance and therapy. Swiss Med Wkly, 2015. 145: p. w14196.
- Medicine, I.o., Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. 2001, Washington DC: National Academy Press.
- Sim, M., et al., Iron considerations for the athlete: a narrative review. Eur J Appl Physiol, 2019. 119(7): p. 1463-1478.
- DellaValle, D.M., Iron supplementation for female athletes: effects on iron status and performance outcomes. Curr Sports Med Rep, 2013. 12(4): p. 234-9.
- Hurrell, R. and I. Egli, Iron bioavailability and dietary reference values. Am J Clin Nutr, 2010. 91(5): p. 1461S-1467S.
- Clarys, P., et al., Comparison of nutritional quality of the vegan, vegetarian, semi-vegetarian, pesco-vegetarian and omnivorous diet. Nutrients, 2014. 6(3): p. 1318-1332.
- Haider, L.M., et al., The effect of vegetarian diets on iron status in adults: A systematic review and metaanalysis. Crit Rev Food Sci Nutr, 2018. 58(8): p. 1359-1374.
- Henjum, S., et al., Iron Status of Vegans, Vegetarians and Pescatarians in Norway. Biomolecules, 2021. 11(3).
- Pawlak, R., J. Berger, and I. Hines, Iron Status of Vegetarian Adults: A Review of Literature. Am J Lifestyle Med, 2018. 12(6): p. 486-498.
- King, J.C., Zinc: an essential but elusive nutrient. The American journal of clinical nutrition, 2011. 94(2): p. 679S-684S.
- King, J.C., Zinc: an essential but elusive nutrient. Am J Clin Nutr, 2011. 94(2): p. 679S-84S.
- Hunt, J.R., Bioavailability of iron, zinc, and other trace minerals from vegetarian diets. The American journal of clinical nutrition, 2003. 78(3): p. 633S-639S.
- Lönnerdal, B., Zinc and health: current status and future directions. J Nutr, 2000. 130: p. 1378-83.
- Fuhrman, J. and D.M. Ferreri, Fueling the vegetarian (vegan) athlete. Current Sports Medicine Reports, 2010. 9(4): p. 233-241.
- Medicine, I.o., Dietary Reference Intakes for Calcium and Vitamin D 2011, Washington DC: National Academy of Sciences.
- Knechtle, B., et al., Vitamin D and Stress Fractures in Sport: Preventive and Therapeutic Measures-A Narrative Review. Medicina (Kaunas), 2021. 57(3).
- Davey, G.K., et al., EPIC–Oxford: lifestyle characteristics and nutrient intakes in a cohort of 33 883 meat-eaters and 31 546 non meat-eaters in the UK. Public health nutrition, 2003. 6(3): p. 259-268.
- Thorpe, D.L., et al., Dietary patterns and hip fracture in the Adventist Health Study 2: combined vitamin D and calcium supplementation mitigate increased hip fracture risk among vegans. Am J Clin Nutr, 2021.
- Maughan, R.J., et al., IOC consensus statement: dietary supplements and the high-performance athlete. International journal of sport nutrition and exercise metabolism, 2018. 28(2): p. 104-125.
- DeLuca, H.F., Overview of general physiologic features and functions of vitamin D. The American journal of clinical nutrition, 2004. 80(6): p. 1689S-1696S.
- Larson-Meyer, D.E., Vegetarian and Vegan Diets for Athletic Training and Performance. Sports Science Exchange, 2018. 29(188): p. 1-7.
- Holick, M.F., Vitamin D deficiency. New England Journal of Medicine, 2007. 357(3): p. 266-281.
- Chan, J., K. Jaceldo-Siegl, and G.E. Fraser, Serum 25-hydroxyvitamin D status of vegetarians, partial vegetarians, and nonvegetarians: the Adventist Health Study-2. The American journal of clinical nutrition, 2009. 89(5): p. 1686S-1692S.
- Sato, T., N. Inaba, and T. Yamashita, MK-7 and Its Effects on Bone Quality and Strength. Nutrients, 2020. 12(4).
- Nakagawa, K., et al., Identification of UBIAD1 as a novel human menaquinone-4 biosynthetic enzyme. Nature, 2010. 468(7320): p. 117-21.
- Nebl, J., et al., Micronutrient Status of Recreational Runners with Vegetarian or Non- Vegetarian Dietary Patterns. Nutrients, 2019. 11(5).
- Brosnan, J.T. and M.E. Brosnan, The sulfurcontaining amino acids: an overview. J Nutr, 2006. 136(6 Suppl): p. 1636S-1640S.
- Carvalho, M.B., et al., Taurine Supplementation Increases Post-Exercise Lipid Oxidation at Moderate Intensity in Fasted Healthy Males. Nutrients, 2020. 12(5).
- Rana, S.K. and T.A. Sanders, Taurine concentrations in the diet, plasma, urine and breast milk of vegans compared with omnivores. Br J Nutr, 1986. 56(1): p. 17-27.
- Kreider, R.B., et al., International Society of Sports Nutrition position stand: safety and efficacy of creatine supplementation in exercise, sport, and medicine. J Int Soc Sports Nutr, 2017. 14: p. 18.
- Kaviani, M., K. Shaw, and P.D. Chilibeck, Benefits of Creatine Supplementation for Vegetarians Compared to Omnivorous Athletes: A Systematic Review. Int J Environ Res Public Health, 2020. 17(9).
- Lukaszuk, J.M., et al., Effect of creatine supplementation and a lacto-ovo-vegetarian diet on muscle creatine concentration. Int J Sport Nutr Exerc Metab, 2002. 12(3): p. 336-48.
- Roschel, H., et al., Creatine Supplementation and Brain Health. Nutrients, 2021. 13(2).
- Harris, R.C., et al., Determinants of muscle carnosine content. Amino Acids, 2012. 43(1): p. 5-12.
- Everaert, I., et al., Vegetarianism, female gender and increasing age, but not CNDP1 genotype, are associated with reduced muscle carnosine levels in humans. Amino Acids, 2011. 40(4): p. 1221-9.
- Trexler, E.T., et al., International society of sports nutrition position stand: Beta-Alanine. J Int Soc Sports Nutr, 2015. 12: p. 30.








